快速叶绿素荧光(OJIP)可作为监测植物在非生物胁迫下光合生理状态的有效工具
欢迎点击「汉莎科学仪器」↑关注我们!
摘要
在自然条件下生活的植物会受到许多干扰光合作用过程的不利因素的影响,导致生长、发育和产量的下降。叶绿素a荧光光谱(ChlF)的研究为叶片光化学效率研究提供了一条新的途径。具体地说,对荧光信号的分析可获取PSII反应中心、捕光天线复合体以及PSII供体侧/受体侧的状态和功能的详细信息。在这里,我们回顾了快速ChlF技术(OJIP & JIP-test)分析光合反应对环境胁迫的相关成果,并讨论了这一创新方法的潜在科学和实际应用。最近便携式设备(Handy PEA & M-PEA, Hansatech Instruments)的出现,特别是在作物表型分型和监测方面,大大扩展了ChlF技术的潜在应用。
关键词 Chlorophyll fluorescence、JIP-test、Photosynthesis、Photosystem II、Quantum efficiency、Stress detection

缩写
Absorption flux | 吸收通量 | |
Chlorophyll | 叶绿素 | |
Chlorophyll fluorescence | 叶绿素荧光 | |
Cross section of the sample | 样品横截面 | |
Cytochrome b6f | 细胞色素b6f | |
Delayed (chlorophyll) fluorescence | 延迟(叶绿素)荧光 | |
Drought factor index | 干旱因子指数 | |
Light-harvesting complex (of PSII) | PSII捕光色素复合体 | |
Oxygen-evolving complex | 放氧复合体 | |
Excited PSII reaction center | 激发的PSII反应中心 | |
PSI reaction center | PSI反应中心 | |
Photosynthetically active radiation | 光合有效辐射 | |
Plastocyanin | 质体蓝素 | |
Principal component analysis | 主成分分析 | |
Prompt (chlorophyll) fluorescence | 瞬时(叶绿素)荧光 | |
Pheophytin | 去镁叶绿素 | |
Plastoquinone | 质体醌 | |
Photosystem I, II | 光系统I, II | |
Primary plastoquinone electron acceptor of PSII | PSII初级质体醌电子受体 | |
Secondary plastoquinone electron acceptor | 次级质体醌电子受体 | |
Reaction center | 反应中心 | |
Reactive oxygen species | 活性氧 |
在自然条件下,植物受到许多不利的环境胁迫因子的影响。这些会破坏光合器官,导致植物生产力和总产量下降。光合作用对环境胁迫特别敏感(Kalaji et al. 2012),使光合测量成为植物胁迫研究的重要组成部分。然而,传统的方法,甚至是技术上先进的方法,如通过气体交换(CO2、H2O和O2)测量光合速率,需要耗费大量时间和人力,且提供的有关整体光合功能的信息并不完整。相比之下,ChlF测量是一种简单、无损、廉价和快速的工具,可用于分析光依赖性光合反应和间接评估同一样本组织中的叶绿素含量(Govindjee 1995; Papageorgiou & Govindjee 2011; Stirbet & Govindjee 2011, 2012)。ChlF方法的这些技术优势使其成为植物育种家(例如作物表型和监测)、生物技术学家、植物生理学家、林业工作者、生态学家和环境学家的流行技术。

关键的是,从植物胁迫研究的角度来看,ChlF测量还提供了有关植物生理状况的间接信息。通过分析叶绿素荧光(ChlF)诱导曲线,可以评估光系统II(PSII)和光合电子传递链的生理状况。它还提供了光依赖的光化学反应和光无关的生化反应的相关信息。总的来说,ChlF测量直接或间接地与依赖光的光合反应的所有阶段有关,包括水的光解、电子传递、类囊体膜上pH梯度的形成、ATP合成以及光合机构的一般生物能条件等(Bernát et al. 2012)。
暗适应叶片照光后可获得多相叶绿素荧光诱导曲线(O–J–I–P-瞬变)(图1)。曲线的轨迹提供了有关光合机构结构和功能的大量信息(Kautsky & Hirsch 1931; Schreiber et al. 1994)。
JIP-test是基于多相快速叶绿素荧光的上升阶段,用于研究光依赖性反应与ChlF的相关性。它基于类囊体膜的“能量流”理论(Strasser et al. 2000)。这个理论可以用简单的代数方程来计算,代表每一个被检测的捕光复合体的总能量流入和流出之间的平衡,并提供关于吸收能量的可能分配的信息。利用这些方程,可以描述PSII复合体之间的能量通信(也称为“聚集grouping”或“连通性connectivity”和“总体分组概率overall grouping probability”)(Stirbet 2013)。
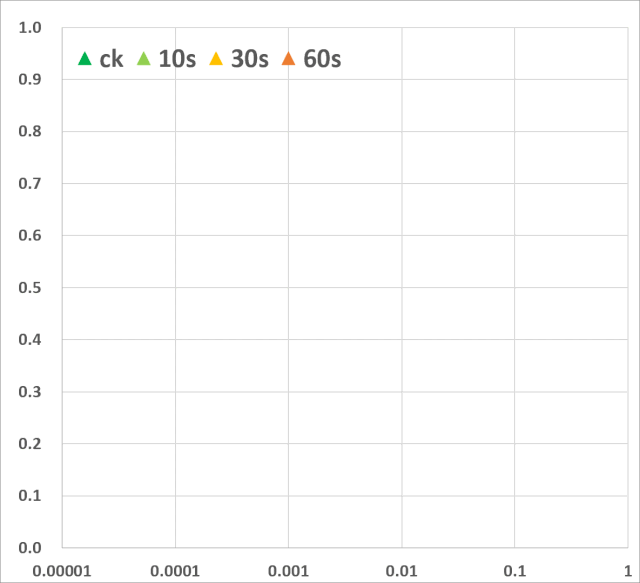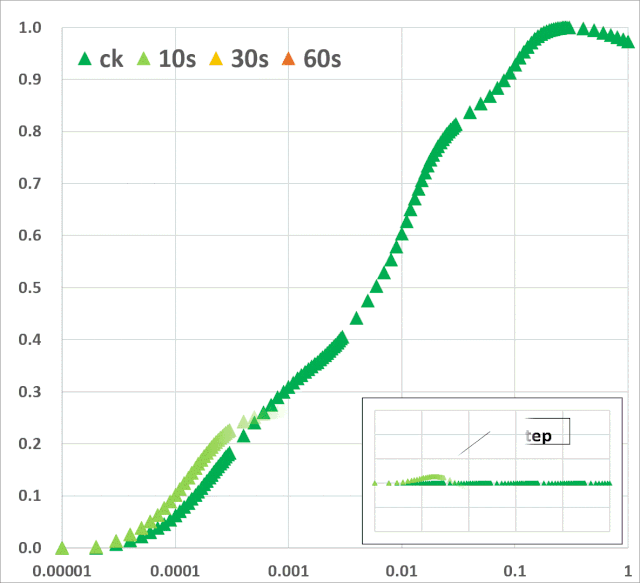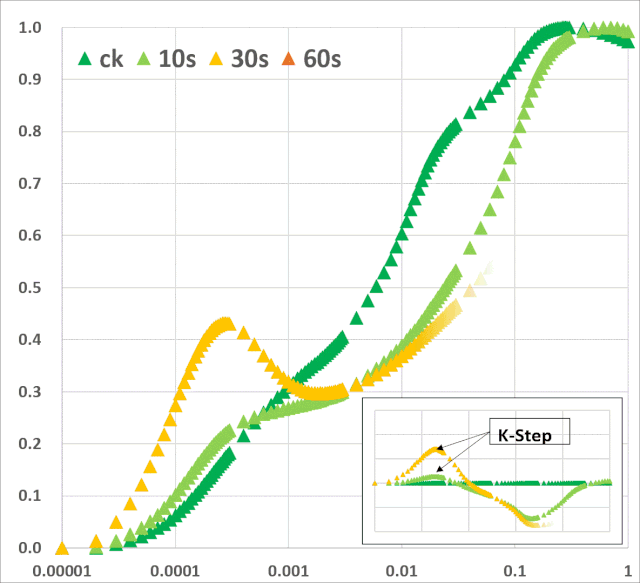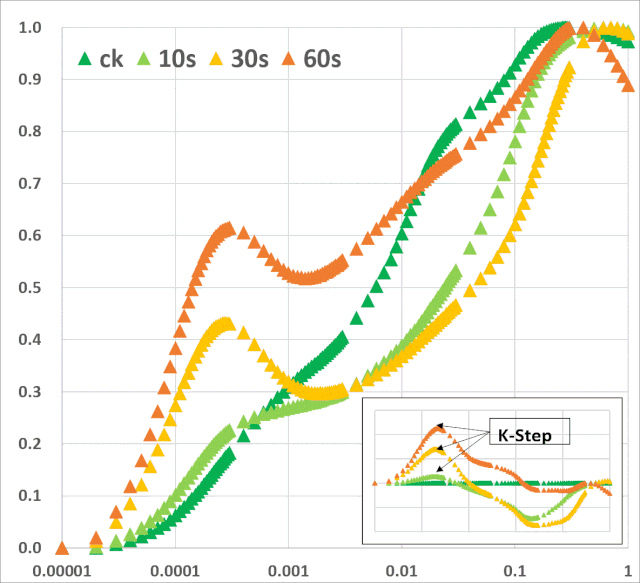
JIP-test(OJIP)的名称来源于ChlF信号形成的感应曲线上的特定位点(图1):这些位点对应于PSII原初电子受体(Pheo)和QA的逐渐还原。诱导曲线的形状取决于PSII各组分间的聚集性(L-band)(Tsimilli-Michaeland Strasser 2013)和电子供体OEC→P680+以及QA-电子的接收之间的平衡(K-band)(Strasser et al. 2005)。
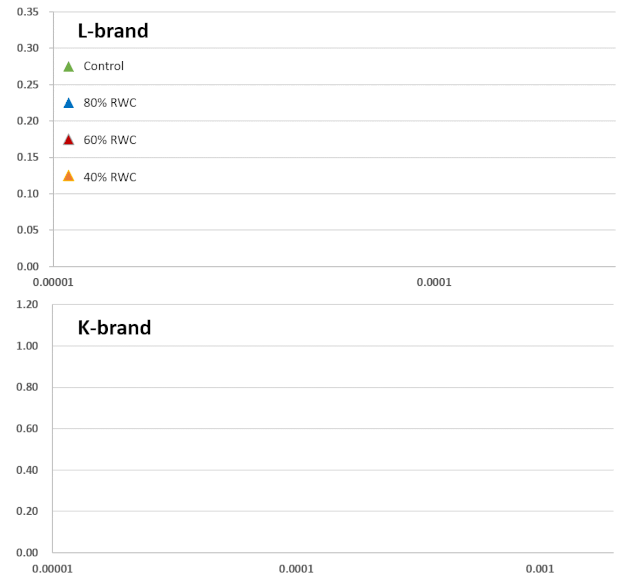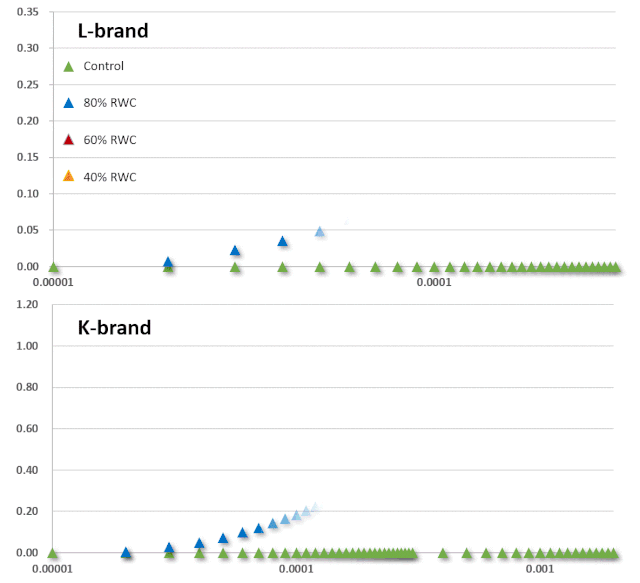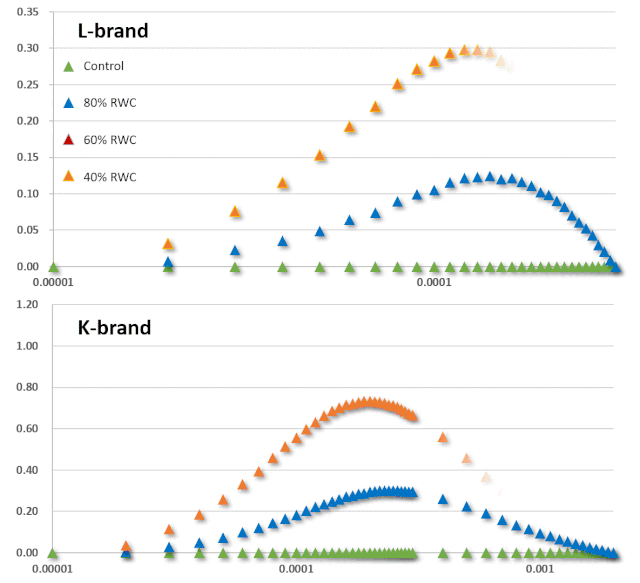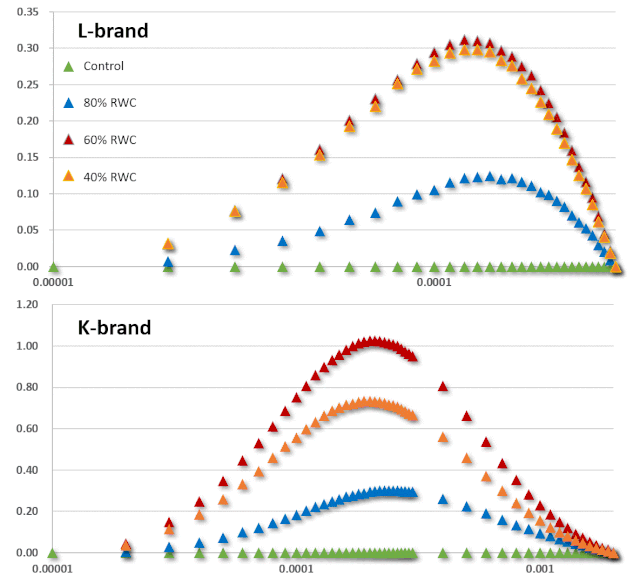
O~J相的荧光上升阶段与部分PSII反应中心的闭合相关,反应了QA的还原水平,其还原程度取决于捕获速率以及QA被QB和其余电子传递链成员氧化的速率。
诱导曲线的J~I相与次级电子受体QB、PQ、Cyt b6f和PC的还原程度相关。诱导曲线的I~P相的上升通常归因于PSI受体侧电子受体(铁氧还原蛋白、中间受体和NADP)的还原。
图1:典型的植物叶绿素荧光多相动力学曲线(主图),曲线以对数时间刻度(10μs~600s)绘制。左上部插图显示了按常规时间标度绘制的相同曲线。右下方插图按常规时间标度绘制的OJIP瞬态(0-30ms)的初始部分。时间标记是指JIP-test用于计算结构和功能参数的选定时间点。
图2:不同胁迫条件下小麦(Triticum sp.L.)叶绿素荧光的O(K)JIP瞬态与非胁迫下的比较。插入显示了O-J相(∆VOJ)、J-I相(∆VJI)、I-P相(∆VIP)的相对可变荧光振幅的变化,以及0.3 ms可变荧光(VK/VJ)与2ms可变荧光比值(VJ)的变化,作为PSII供体侧限制(K-band)的指标。各图显示了相对于非胁迫状态下植物(control,C)的瞬时荧光曲线:a热胁迫(高温胁迫8h,中度光化光照射,叶片温度约40℃);b低温胁迫10d(10/6℃:日间/夜间);c重度干旱胁迫(停止灌溉后12d,叶片含水量约60%);d盐胁迫(NaCl);e氮缺乏胁迫(低氮,LN);f铅胁迫。
AntosiewiczDM (2005) Study of calcium-dependent lead-tolerance on plants differing intheir level of Ca-deficiency tolerance. Environ Pollut 134(1):23–34 BeddingtonJ, Asaduzzaman M, Clark M, Bremauntz AF, Guillou M, Howlett D, Jahn M, Lin E,Mamo T, Negra C (2012) What next for agriculture after Durban? Science335:289–290 BernátG, Schreiber U, Sendtko E, Stadnichuk IN, Rexroth S, Rögner M, Koenig F (2012)Unique properties vs. common themes: the atypical cyanobacterium Gloeobacterviolaceus PCC 7421 is capable of state transitions and blue-light-inducedfluorescence quenching. Plant Cell Physiol 53(3):528–542 BoureimaS, Oukarroum A, Diouf M, Cisse N, Van Damme P (2012) Screening for droughttolerance in mutant germplasm of sesame (Sesamum indicum) probing by chlorophylla fluorescence. Environ Exp Bot 81:37–43 BresticM, Zivcak M (2013) PSII Fluorescence techniques for measurement of drought andhigh temperature stress signal in crop plants: protocols and applications.Molecular stress physiology of plants. Springer, Berlin, pp 87–131 BresticM, Zivcak M, Kalaji MH, Carpentier R, Allakhverdiev SI(2012) Photosystem IIthermostability in situ: environmentally induced acclimation andgenotype-specific reactions in Triticum aestivum L. Plant PhysiolBiochem 57:93–105 BresticM, Zivcak M, Olsovska K, Repkova J (2013) Involvement of chlorophyll afluorescence analyses for identification of sensitiveness of the photosyntheticapparatus to high temperature in selected wheat genotypes. Photosynthesisresearch for food, fuel and the future. Springer, Berlin, pp 510–513 BresticM, Zivcak M, Olsovska K, Shao HB, Kalaji MH, Allakhverdiev SI (2014) Reducedglutamine synthetase activity plays a role in control of photosyntheticresponses to high light in barley leaves. Plant Physiol Biochem 81:74–83.doi:10.1016/j.plaphy.2014.01.002 CeppiMG, Oukarroum A, C ¸i c ¸ek N, Strasser RJ, Schansker G (2012) The IP amplitudeof the fluorescence rise OJIP is sensitive to changes in the photosystem Icontent of leaves: a study on plants exposed to magnesium and sulfatedeficiencies, drought stress and salt stress. Physiol Plant 144(3):277–288 ChavesMM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress:regulation mechanisms from whole plant to cell. Ann Bot 103(4):551–560 ChenLS, Li P, Cheng L (2009) Comparison of thermotolerance of sun-exposed peel andshaded peel of ‘Fuji’apple. Environ Exp Bot 66(1):110–116 CorreiaCM, Pereira JMM, Coutinho JF, Björn LO, Torres-Pereira JMG (2005) Ultraviolet-Bradiation and nitrogen affect the photosynthesis of maize: a Mediterraneanfield study. Eur J Agron 22(3):337–347 DudejaSS, Chaudhary P (2005) Fast chlorophyll fluorescence transient and nitrogenfixing ability of chickpea nodulation variants. Photosynthetica 43:253–259 Ekmekc¸i Y, Tanyolac D, Ayhan B (2008) Effects of cadmium on antioxidant enzyme andphotosynthetic activities in leaves of two maize cultivars. J Plant Physiol165(6):600–611 EvansJR (2009) Potential errors in electron transport rates calculated fromchlorophyll fluorescence as revealed by a multilayer leaf model. Plant CellPhysiology 50(4):698–706 FoyerC, Spencer C (1986) The relationship between phosphate status andphotosynthesis in leaves. Planta 167(3):369–375 GautumA, Agrawal D, SaiPrasad SV, Jajoo A (2014) A quick method to screen high andlow yielding wheat cultivars exposed to high temperature. Physiol Mol BiolPlants. doi:10.1007/s12298-014-0252-4 GoltsevV, Zaharieva I, Chernev P, Kouzmanova M, Kalaji MH, Yordanov I, Krasteva V,Alexandrov V, Stefanov D, Allakh-verdiev SI, Strasser RJ (2012) Drought-inducedmodifications of photosynthetic electron transport in intact leaves: analysisand use of neural networks as a tool for a rapid non-invasive estimation.Biochim Biophys Acta 1817:1490–1498 GoltsevV, Zaharieva I, Chernev P, Strasser RJ (2009) Delayed chlorophyll fluorescenceas a monitor for physiological state of photosynthetic apparatus. BiotechnolBiotechnol Equip 23(Special Edition):452–457 GomesMTG, da Luz AC, dos Santos MR, Batitucci MDCP, Silva DM, Falqueto AR (2012)Drought tolerance of passion fruit plants assessed by the OJIP chlorophyll afluorescence transient. Sci Hortic 142:49–56 GottardiniE, Cristofori A, Cristofolini F, Nali C, Pellegrini E, Bussotti F, Ferretti M(2014) Chlorophyll-related indicators are linked to visible ozone symptoms:evidence from a field study on native Viburnum lantana L. plants innorthern Italy. Ecol Ind 39:65–74 Govindjee(1995) Sixty-three years since Kautsky: chlorophyll a fluorescence. AusJ Plant Physiol 22:131–160 GuanterL, Zhang Y, Jung M, Joiner J, Voigt M, Berry JA, Frankenberg C, Huete AR,Zarco-Tejada P, Lee JE (2014) Global and time-resolved monitoring of cropphotosynthesis with chlorophyll fluorescence. Proc Natl Acad Sci111(14):E1327–E1333 GuhaA, Sengupta D, Reddy AR (2013) Polyphasic chlorophyll a fluorescencekinetics and leaf protein analyses to track dynamics of photosyntheticperformance in mulberry during progressive drought. J Photochem Photobiol B119:71–83 GuisséB, Srivastava A, Strasser RJ (1995) Effects of high temperature and waterstress on the polyphasic chlorophyll a fluorescence transient of potatoleaves. In: Mathis P (ed) Photosynthesis: from light to biosphere. KluwerAcademic Publishers, Dordrecht, pp 913–916 HavauxM (1993) Rapid photosynthetic adaptation to heat stress triggered in potatoleaves by moderately elevated temperatures. Plant Cell Environ 16(4):461–467 HeY, Zhu Z, Yang J, Ni X, Zhu B (2009) Grafting increases the salt tolerance oftomato by improvement of photosynthesis and enhancement of antioxidant enzymesactivity. Environ Exp Bot 66(2):270–278 HuangZA, Jiang DA, Yang Y, Sun JW, Jin SH (2004) Effects of nitrogen deficiency ongas exchange, chlorophyll fluorescence, and antioxidant enzymes in leaves ofrice plants. Photosynthetica 42(3):357–364 JajooA (2013) Changes in photosystem II in response to salt stress. Ecophysiologyand responses of plants under salt stress. Springer, Berlin, pp 149–168 JaneczkoA, Koscielniak J, Pilipowicz M, Szarek-Lukaszewska G, Skoczowski A (2005)Protection of winter rape photosystem 2 by 24-epibrassinolide under cadmiumstress. Photosynthetica 43(2):293–298 JedmowskiC, Ashoub A, Bru ¨ggemann W (2013) Reactions of Egyptian landraces of Hordeumvulgare and Sorghum bicolor to drought stress, evaluated by the OJIP fluorescencetransient analysis. Acta Physiologiae Plantarum 35(2):345–354 KalajiMH, Rutkowska A (2004) Reactions of photosynthetic apparatus of maize seedlingsto salt stress. Zesz Probl Post NaukRol 496:545–558 KalajiMH, Carpentier R, Allakhverdiev SI, Bosa K (2012) Fluorescence parameters asearly indicators of light stress in barley. J Photochem Photobiol B 112:1–6 KalajiMH, Schansker G, Ladle RJ, et al (2014a) Frequently asked questions about invivo chlorophyll fluorescence: practical issues. Photosynth Res. doi:10.1007/s11120-014-0024-6 KalajiMH, Oukarroum A, Alexandrov V, Kouzmanova M, Brestic M, Zivcak M, Goltsev V(2014b) Identification of nutrient deficiency in maize and tomato plants by invivo chlorophyll a fluorescence measurements. Plant Physiol Biochem.doi:10.1016/j.plaphy.2014.03.029 KautskyH, Hirsch A (1931) NeueVersuchezur Kohlensaureassimilation. Naturwissenschaften19:964 KrugerGHJ, Tsimilli-Michael M, Strasser RJ (1997) Light stress provokes plastic andelastic modifications in structure and function of photosystem II in camellialeaves. Physiol Plant 101:265–277 LaurianoJA, Ramalho JC, Lidon FC, Ce úmatos M (2006) Mechanisms of energy dissipationin peanut under water stress. Photosynthetica 44(3):404–410 LazárD (2006) The polyphasic chlorophyll a fluorescence rise measured under highintensity of exciting light. Funct Plant Biol 33(1):9–30 LazárD, Jablonsky J (2009) On the approaches applied in formulation of a kineticmodel of photosystem II: different approaches lead to different simulations ofthe chlorophyll a fluorescence transients. J Theor Biol 257(2):260–269 LiG, Zhang ZS, Gao HY, Liu P, Dong ST, Zhang JW, Zhao B (2012) Effects ofnitrogen on photosynthetic characteristics of leaves from two differentstay-green corn (Zea mays L.) varieties at the grain-filling stage. CanJ Plant Sci 92:671–680 LiuWJ, Chen YE, Tian WJ, Du JB, Zhang ZW, Xu F, Zhang F, Yuan S, Lin HH (2009)Dephosphorylation of photosystem II proteins and phosphorylation of CP29 inbarley photosynthetic membranes as a response to water stress. Biochim BiophysActa 1787(10):1238–1245 LobellDB, Burke MB, Tebaldi C, Mastrandrea MD, Falcon WP, Naylor RL (2008)Prioritizing climate change adaptation needs for food security in 2030. Science319:607–610 ŁobodaT, Kalaji MH (2007) Photosystem II of barley seedlings under cadmium and leadstress. Plant Soil Environ 53(12):511 MalaspinaP, Giordani P, Faimali M, Garaventa F, Modenesi P (2014) Assessingphotosynthetic biomarkers in lichen transplants exposed under different lightregimes. Ecol Ind 43:126–131 MallickN, Mohn F (2003) Use of chlorophyll fluorescence in metalstress research: acase study with the green microalga Scenedesmus. Ecotoxicol Environ Saf55(1):64–69 MathurS, Mehta P, Jajoo A, Bharti S (2011a) Analysis of elevated temperature inducedinhibition of Photosystem II using Chl a fluorescence inductionkinetics. Plant Biology 13:1–6 MathurS, Allakhverdiev SI, Jajoo A (2011b) Analysis of high temperature stress on thedynamics of antenna size and reducing side heterogeneity of Photosystem II inwheat leaves (Triticum aestivum). Biochim Biophys Acta 1807(1):22–29 MathurS, Agrawal D, Jajoo A (2014) Photosynthesis: limitations in response to hightemperature stress. J Photochem Photobiol B Biol. doi: 10.1016/j.jphotobiol.2014.01.010 MehtaP, Allakhverdiev SI, Jajoo A (2010a) Characterization of photosystem IIheterogeneity in response to high salt stress in wheat leaves (Triticumaestivum). Photosynth Res 105(3):249–255 MehtaP, Jajoo A, Mathur S, Bharti S (2010b) Chlorophyll a fluorescence studyrevealing effects of high salt stress on Photosystem II in wheat leaves. PlantPhysiol Biochem 48(1):16–20 MelisA, Homann PH (1976) Heterogeneity of the photochemical centers in system II ofchloroplasts. Photochem Photobiol 23:343–350 MishraS, Dubey R (2005) Heavy metal toxicity induced alterations in photosyntheticmetabolism in plants. In: Pessarakli M (ed) Handbook of photosynthesis, vol 28.CRC Press, Boca Raton, pp 827–844 MolassiotisA, Tanou G, Diamantidis G, Patakas A, Therios I (2006) Effects of 4-month Fedeficiency exposure on Fe reduction mechanism, photosynthetic gas exchange,chlorophyll fluorescence and antioxidant defense in two peach rootstocksdiffering in Fe deficiency tolerance. J Plant Physiol 163(2):176–185 MurataN, Mohanty P, Hayashi H, Papageorgiou G (1992) Glycinebetaine stabilizes theassociation of extrinsic proteins with the photosynthetic oxygen-evolvingcomplex. FEBS Lett 296(2):187–189 OukarroumA, Madidi SE, Schansker G, Strasser RJ (2007) Probing the responses of barleycultivars (Hordeum vulgare L.) by chlorophyll a fluorescence OLKJIPunder drought stress and rewatering. Environ Exp Bot 60(3):438–446 OukarroumA, Schansker G, Strasser RJ (2009) Drought stress effects on photosystem Icontent and photosystem II thermotolerance analyzed using Chl afluorescence kinetics in barley varieties differing in their drought tolerance.Physiol Plant 137(2):188–199 OukarroumA, El Madidi S, Strasser RJ (2012) Exogenous glycine betaine and proline play aprotective role in heat-stressed barley leaves (Hordeum vulgare L.): achlorophyll a fluorescence study. Plant Biosyst 146(4):1037–1043 PapageorgiouGC, Govindjee (2011) Photosystem II fluorescence: slow changes—scaling from thepast. J Photochem Photobiol B 104(1–2):258–270 RampinoP, Pataleo S, Gerardi C, Mita G, Perrotta C (2006) Drought stress response inwheat: physiological and molecular analysis of resistant and sensitivegenotypes. Plant Cell Environ 29(12):2143–2152 RedillasMCFR, Jeong JS, Strasser RJ, Kim YS, Kim JK (2011) JIP analysis on rice (Oryzasativa cv Nipponbare) grown under limited nitrogen conditions. J Korean SocAppl Biol Chem 54:827–832 RomanowskaE, Igamberdiev AU, Parys E, Gardeström P (2002) Stimulation of respiration byPb2+ in detached leaves and mitochondria of C3 and C4 plants.Physiol Plant 116(2):148–154 RomanowskaE, Wróblewska B, Dro_ zak A, Siedlecka M (2006) High light intensity protects photosyntheticapparatus of pea plants against exposure to lead. Plant Physiol Biochem44(5):387–394 SchanskerG, Srivastava A, Govindjee, Strasse RJ (2003) Characterization of the 820-nmtransmission signal paralleling the chlorophyll a fluorescence rise (OJIP)in pea leaves. Funct Plant Biol 30(7):785–796 SchreiberU, Neubauer C, Klughammer C (1989) Devices and methods for room-temperaturefluorescence analysis. Philos Trans Royal Soc London B Biol Sci323(1216):241–251 SchreiberU, Bilger W, Neubauer C (1994) Chlorophyll fluorescence as a nonintrusiveindicator for rapid assessment of in vivo photosynthesis. Ecophysiology ofphotosynthesis. Springer, Berlin, pp 49–70 SchreiberU, Klughammer C, Kolbowski J (2012) Assessment of wavelength-dependent parametersof photosynthetic electron transport with a new type of multi-color PAMchlorophyll fluorometer. Photosynth Res 113(1–3):127–144 SchweigerJ, Lang M, Lichtenthaler HK (1996) Differences in fluorescence excitationspectra of leaves between stressed and non-stressed plants. J Plant Physiol148(5):536–547 SharkeyTD, Schrader SM (2006) High temperature stress. Physiology and molecularbiology of stress tolerance in plants. Springer, Berlin, pp 101–129 SharmaP, Dubey RS (2005) Lead toxicity in plants. Brazilian Journal of PlantPhysiology 17:35–52 SigfridssonKG, Berna ′t G, Mamedov F, Styring S (2004) Molecular interference of Cd2+with Photosystem II. Biochim Biophys Acta 1659(1):19–31 Singh-TomarR, Mathur S, Allakhverdiev SI, Jajoo A (2012) Changes in PSII heterogeneity inresponse to osmotic and ionic stress in wheat leaves (Triticum aestivum). JBioenerg Biomembr 44:411–419 SmethurstCF, Garnett T, Shabala S (2005) Nutritional and chlorophyll fluorescenceresponses of lucerne (Medicago sativa) to waterlogging and subsequent recovery.Plant Soil 270(1):31–45 SrivastavaA, Strasser RJ (1995) How do land plants respond to stress temperature andstress light? Arch Sci Geneve 48:135–146 SrivastavaA, Strasser RJ, Govindjee (1999) Greening of peas: parallel measurements of 77K emission spectra, OJIP chlorophyll a fluorescence transient, periodfour oscillation of the initial fluorescence level, delayed light emission, andP700. Photosynthetica 37:365–392 StefanovD, Petkova V, Denev ID (2011) Screening for heat tolerance in common bean (Phaseolusvulgaris L.) lines and cultivars using JIP-test. Sci Hortic 128(1):1–6 StirbetA (2013) Excitonic connectivity between photosystem II units: what is it, andhow to measure it? Photosynth Res 116:189–214 StirbetA, Govindjee (2011) On the relation between the Kautsky effect (chlorophyll afluorescence induction) and Photosystem II: basics and applications of the OJIPfluorescence transient. J Photochem Photobiol B 104(1–2):236–257 StirbetA, Govindjee (2012) Chlorophyll a fluorescence induction: a personalperspective of the thermal phase, the J–I–P rise. Photosynth Res 113:15–61 StrasserRJ, Stirbet AD (1998) Heterogeneity of photosystem II probed by the numericallysimulated chlorophyll a fluorescence rise (O–J–I–P). Math Comput Simul48(1):3–9 StrasserRJ, Srivastava A, Tsimilli-Michael M (2000) The fluorescence transient as atool to characterize and screen photosynthetic samples. In: Mohanty P, Yunus,Pathre (eds) Probing photosynthesis: mechanism, regulation and adaptation.Taylor and Francis, London, pp 443–480 StrasserRJ, Tsimilli-Michael M, Srivastava A (2004) Analysis of the chlorophyll afluorescence transient. In: Papageorgiou G, Govindjee (eds) Advances inphotosynthesis and respiration. chlorophyll a fluorescence: a signatureof photosynthesis. Springer, Dordrecht, pp 321–362 StrasserRJ, Tsimilli-Michael M, Srivastava A, Srivastava A (2005) Analysis of thechlorophyll a fluorescence transient. In: Papageorgiou GC, Govindjee(eds) Advances in photosynthesis and respiration. Chlorophyll afluorescence: a signature of photosynthesis. Kluwer Acad. Publ, Dordrecht, pp321–362 StrasserRJ, Tsimilli-Michael M, Qiang S, Goltsev V (2010) Simultaneous in vivorecording of prompt and delayed fluorescence and 820-nm reflection changesduring drying and after rehydration of the resurrection plant Haberlearhodopensis. Biochimical BiophysicalActa 1797:1313–1326 StraussA, Kru ¨ger G, Strasser RJ, Heerden PV (2006) Ranking of dark chillingtolerance in soybean genotypes probed by the chlorophyll a fluorescencetransient OJIP. Environ Exp Bot 56(2):147–157 StrebP, Aubert S, Gout E, Feierabend J, Bligny R (2008) Cross tolerance toheavy-metal and cold-induced photoinhibiton in leaves of Pisum sativumacclimated to low temperature. Physiol Mole Biol Plants 14(3):185–193 SuzukiK, Ohmori Y, Ratel E (2011) High root temperature blocks both linear and cyclicelectron transport in the dark during chilling of the leaves of rice seedlings.Plant Cell Physiology 52(9):1697–1707 TomarRS, Jajoo A (2013) A quick investigation of the detrimental effects ofenvironmental pollutant polycyclic aromatic hydrocarbon fluoranthene on thephotosynthetic efficiency of wheat (Triticum aestivum). Ecotoxicology22(8):1313–1318 TomarRS, Jajoo A (2014) Fluoranthene, a polycyclic aromatic hydrocarbon, inhibitslight as well as dark reactions of photosynthesis in wheat (Triticum aestivum).Ecotoxicol Environ Saf 109:110–115 Tsimilli-MichaelM, Strasser RJ (2008) In vivo assessment of stress impact on plants’ vitality:applications in detecting and evaluating the beneficial role of Mycorrhizationon host plants. In: Varma A (ed) Mycorrhiza: state of the art, genetics and molecularbiology, eco-function, biotechnology, eco-physiology, structure andsystematics, vol 3rd. Springer, Berlin, pp 679–703 Tsimilli-MichaelM, Strasser RJ (2013) The energy flux theory 35 years later: formulations andapplications. Photosynth Res 117(1–3):289–320. doi:10.1007/s11120-013-9895-1 TubaZ, Saxena DK, Srivastava K, Singh S, Czebol S, Kalaji MH (2010) Chlorophyll afluorescence measurements for validating the tolerant bryophytes for heavymetal (Pb) biomapping. Curr Sci 98(11):1505–1508 VanHeerden PD, Strasser RJ, Kru ¨ger GH (2004) Reduction of dark chilling stressin N2-fixing soybean by nitrate as indicated by chlorophyll a fluorescencekinetics. Physiol Plant 121:239–249 VanHeerden PDR, Swanepoel JW, Kru ¨ger GHJ (2007) Modulation of photosynthesis bydrought in two desert scrub species exhibiting C3-mode CO2 assimilation.Environ Exp Bot 61(2):124–136 WuC, Wang Z, Sun H, Guo S (2006) Effects of different concentrations of nitrogenand phosphorus on chlorophyll biosynthesis, chlorophyll a fluorescence,and photosynthesis in Larix olgensis seedlings. Front Forest China1(2):170–175 WuX, Hong F, Liu C, Su M, Zheng L, Gao F, Yang F (2008) Effects of Pb2+on energy distribution and photochemical activity of spinach chloroplast.Spectrochimica Acta Part A Mol Biomol Spectrosc 69(3):738–742 YamaneY, Kashino Y, Koike H, Satoh K (1997) Increases in the fluorescence Fo leveland reversible inhibition of Photosystem II reaction center by high-temperaturetreatments in higher plants. Photosynth Res 52:57–64 YangX, Liang Z, Wen X, Lu C (2008) Genetic engineering of the biosynthesis ofglycinebetaine leads to increased tolerance of photosynthesis to salt stress intransgenic tobacco plants. Plant Mol Biol 66(1–2):73–86 YangJ, Kong Q, Xiang C (2009) Effects of low night temperature on pigments, chl afluorescence and energy allocation in two bitter gourd (Momordica charantiaL.) genotypes. Acta Physiologiae Plantarum 31(2):285–293 ZhangR, Sharkey TD (2009) Photosynthetic electron transport and proton flux undermoderate heat stress. Photosynth Res 100(1):29–43 ZivcakM, Brestic M, Olsovska K, Slamka P (2008) Performance index as a sensitiveindicator of water stress in Triticum aestivum. Plant Soil Environ54:133–139 ZivcakM, Brestic M, Balatova Z, Drevenakova P, Olsovska K, Kalaji MH, AllakhverdievSI (2013) Photosynthetic electron transport and specific photoprotectiveresponses in wheat leaves under drought stress. Photosynth Res 117:529–546 ZivcakM, Kalaji MH, Shao HB, Olsovska K, Brestic M (2014). Photosynthetic proton andelectron transport in wheat leaves under prolonged moderate drought stress. JPhotochem Photobiol B Biol. doi: 10.1016/j.jphotobiol.2014.01.007 ZivcakM, Olsovska K, Slamka P, Galambosova J, Rataj V, Shao HB, Brestic M (2014b)Application of chlorophyll fluorescence performance indices to assess the wheatphotosynthetic functions influenced by nitrogen deficiency. Plant Soil Environ60:210–215 ZushiK, Kajiwara S, Matsuzoe N (2012) Chlorophyll a fluorescence OJIP transient as atool to characterize and evaluate response to heat and chilling stress intomato leaf and fruit. Sci Hortic 148:39–46
推荐
-

-

QQ空间
-

新浪微博
-

人人网
-

豆瓣

